 
Once this happens, the salt is dissolved, resulting in a homogeneous solution. Ionic liquids (ILs) are liquid organic salts possessing a lot of interesting properties that prompt various researchers to investigate their utilization in various fields 1, 2, 3, 4. Because of their outstanding versatility, room-temperature ionic liquids (RTILs) are utilized in an ever increasing number of novel and fascinating applications, making them the Holy Grail of modern materials science. After the salt compounds are pulled apart, the sodium and chloride atoms are surrounded by water molecules, as this diagram shows. Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together. Essentially, a tug-of-war ensues with the water molecules winning the match. The positively-charged side of the water molecules are attracted to the negatively-charged chloride ions and the negatively-charged side of the water molecules are attracted to the positively-charged sodium ions. ionic bond: electrostatic forces of attraction between the oppositely charged ions of an ionic compound. When salt is mixed with water, the salt dissolves because the covalent bonds of water are stronger than the ionic bonds in the salt molecules. 12 explain why ionic and covalent network substances are solid (high melting points), and why covalent molecular substances can be solid, liquid or gas (low. Likewise, a water molecule is ionic in nature, but the bond is called covalent, with two hydrogen atoms both situating themselves with their positive charge on one side of the oxygen atom, which has a negative charge. The bonds in salt compounds are called ionic because they both have an electrical charge-the chloride ion is negatively charged and the sodium ion is positively charged. Ionic, on the other hand, refers to substances that are made up of ions, which are atoms or molecules that have gained or lost electrons and have an electric charge. 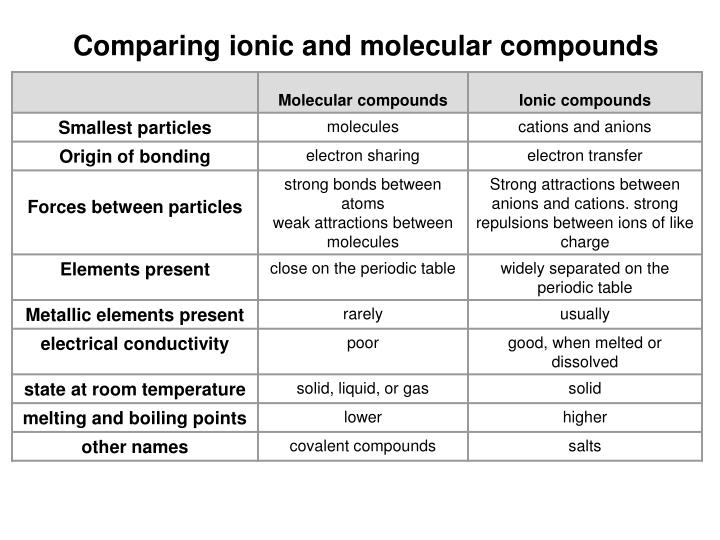
It also depicts how a charge, such as on an ion (Na or Cl, for example) can interact with a water molecule.Īt the molecular level, salt dissolves in water due to electrical charges and due to the fact that both water and salt compounds are polar, with positive and negative charges on opposite sides in the molecule. Molecular refers to substances that are made up of molecules, which are groups of atoms held together by covalent bonds. This diagram shows the positive and negative parts of a water molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
